
Activation of Alcohols as Sulfonium Salts in the Photocatalytic Hetero-Difunctionalization of Alkenes
H. Zhao, D. Filippini, Y. Chen, A. Gallego-Gamo, L. S. Natrajan, L. Pantaine, C. Romano, D. J. Procter* Nat. Chem. 2026, 18, 398–406


Contemporary Strategies in SmI2 Catalysis: a Reagent Reborn
J. I. Mansell, C. Romano, D. J. Procter* Angew. Chem. Int. Ed. 2025, 64, e202519678


Biocatalytic Activation of Sulfur Heteroaromatics Facilitates Dearomatizing Cross-Couplings to Set Stereogenic Centers or Axes
Ó. Conboy, E. Q. Rushworth, C. J. Taylor, C. W. Levy, M. Ortmayer, G. F. S. Whitehead, A. Yen, C. Romano, A. P. Green*, D. J. Procter* J. Am. Chem. Soc. 2025, 147, 43057–43066


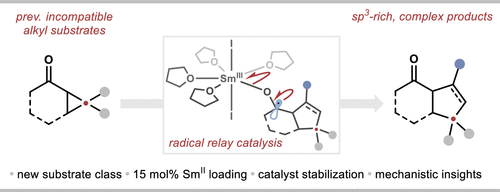
SmI2-Catalyzed Coupling of Alkyl Housane Ketones and Alkenes in an Approach to Norbornanes
D. Roy, J. I. Mansell, G. Barison, S. Yu, R. Katavic, C. Romano, N. Kaltsoyannis, D. J. Procter* Angew. Chem. Int. Ed. 2025, 64, e202512018


Transition Metal-Free Formal C–H/C–H Coupling of Arylacetamides and Sulfoxides: an Interrupted Pummerer/[2,3] Sigmatropic Rearrangement Sequence
S. Zhang, Y. X. Lou, A. Larroza, B. W. Joynson, C. Romano, D. J. Procter* Angew. Chem. Int. Ed. 2025, 64, e202511703


Direct Activation of Sulfides by C–H Oxidation with Photoexcited Nitroarenes: Formal Manipulations of the C–S Bond
V. D. Cuomo, C. Romano*, D. J. Procter* Angew. Chem. Int. Ed. 2025, 64, e202509244


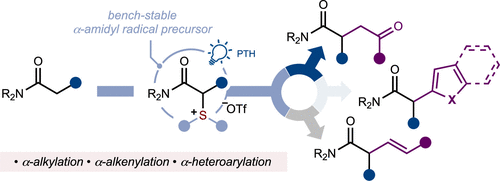
α-Amido Sulfonium Salts Provide a Platform for Photocatalytic Metal-free Carbon-Carbon Bond Formation in Amides
L. Van Dalsen, S. Zhang, W. Tian, B. Joynson, C. Romano, D. J. Procter* ACS Catal. 2025, 15, 8345–8352

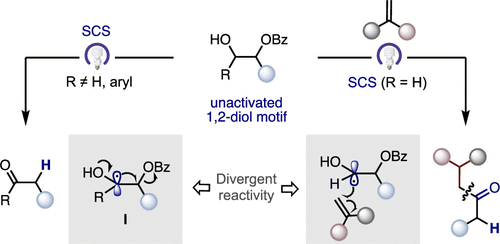
Catalytic Deoxygenation of Unactivated 1,2-Diol Motifs via Light-Induced Spin-Center Shift
J. Ordóñez, A. Brenes Rucinski, C. Romano, R. Martin* ACS Catal. 2025, 15, 3499–3504

Light-Assisted Functionalization Leveraging Aryl Radicals: towards Metal-Free Cross-Coupling
H. Zhao, V. D. Cuomo, W. Tian, C. Romano*, D. J. Procter* Nat. Rev. Chem. 2025, 9, 61–80


Computational Study of SmI2-Catalyzed Intermolecular Couplings of Cyclopropyl Ketones: Links between Structure and Reactivity
S. Yu, C. Romano, D. J. Procter, N. Kaltsoyannis* J. Org. Chem. 2024, 89, 15842–15850

Recent Advances in Ni-Catalyzed Remote C(sp3)–H Functionalization by Chain-Walking Strategies
C. Romano*, R. Martin* Nat. Rev. Chem. 2024, 8, 833–850


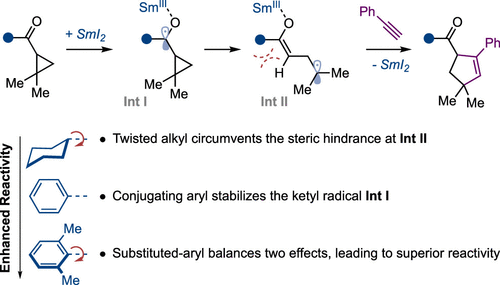
Alkyl Cyclopropyl Ketones in Catalytic Formal [3+2] Cycloadditions: the Role of SmI2 Catalyst Stabilization
J. I. Mansell, S. Yu, M. Li, E. Pye, C. Yin, F. Beltran, J. A. Rossi-Ashton, C. Romano, N. Kaltsoyannis, D. J. Procter* J. Am. Chem. Soc. 2024, 146, 12799–12807

A Blueprint for Catalysis
C. Romano, J. I. Mansell, D. J. Procter* Nat. Chem. 2024, 16, 478


Conformational Flexibility as a Tool for Enabling Site-Selective Functionalization of Unactivated sp3 C–O Bonds in Cyclic Acetals
C. Romano, L. Talavera, E. Gómez-Bengoa, R. Martin* J. Am. Chem. Soc. 2022, 144, 11558–11563


Dual Catalytic Platform for Enabling sp3 α C–H Arylation and Alkylation of Benzamides
A. W. Rand, H. Yin, L, Xu, J. Giacoboni, R. Martin-Montero, C. Romano, J. Montgomery*, R. Martin* ACS Catal. 2020, 10, 4671–4676


Site-Selective Catalytic Deaminative Alkylation of Unactivated Olefins
S. Z. Sun, C. Romano, R. Martin* J. Am. Chem. Soc. 2019, 141, 16197–16201


Remote Functionalization of α,β-Unsaturated Carbonyls by Multimetallic Sequential Catalysis
C. Romano, D. Fiorito, C. Mazet* J. Am. Chem. Soc. 2019, 141, 16983–16990


Multicatalytic Stereoselective Synthesis of Highly Substituted Alkenes by Sequential Isomerization/Cross-Coupling Reactions
C. Romano, C. Mazet* J. Am. Chem. Soc. 2018, 140, 4743–4750


Palladium-catalyzed Long-range Deconjugative Isomerization of Highly Substituted α,β-Unsaturated Carbonyl Compounds
C. Romano, L. Lin, C. Mazet* J. Am. Chem. Soc. 2016, 138, 10344–10350


Metal-free Enantioselective Electrophilic Activation of Allenamides: Stereoselective Dearmatization of Indoles
C. Romano, M. Jia, M. Monari, E. Manoni, M. Bandini* Angew. Chem. Int. Ed. 2014, 53, 13854–13857


Chiral Auxiliary Induced Diatereoselective Synthesis of (R,R)-N, N’-Di(tert-butoxycarbonyl)cyclohex-4-ene-1,2-diamine
D. Balestri, S. Grilli, C. Romano, D. Savoia* Eur. J. Org. Chem. 2014, 8021–8025

